The central nervous system (CNS) consists of neurons and glial cells. Glial cells are astrocytes, oligodendroglia, ependymal cells, and microglia. With H&E stains, the CNS resembles mesenchymal tissues in which cells are set in an extracellular matrix. This is a wrong impression. The fibrillary "matrix" of the cerebral gray matter, the neuropil is formed by the cellular extensions (processes) of the neurons and glial cells. These processes fit together tightly, leaving a minimal extracellular space. The neuropil is traversed by blood vessels. As leptomeningeal vessels penetrate the brain, the subarachnoid space dips into CNS tissue around them, creating a perivascular (Virchow-Robin) space. This is accentuated in paraffin-embedded CNS tissue and appears as an empty space. The Virchow-Robin space extends down to the level of arterioles and venules. In brain capillaries, astrocytes are apposed to the vessel wall.
Neurons
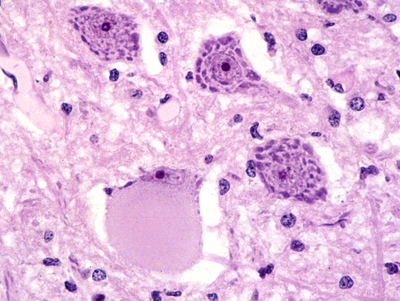
Each neuron has a cell body (the perikaryon), an axon, and dendrites. The dendritic tree is the receptive part of the neuron. The axon conveys the signal to its target. The cell body contains the nucleus and most organelles that perform the synthetic and catabolic activities that keep the neuron and all its parts alive and functioning. Neurons come in all sizes. Motor neurons, which are the largest cells in the CNS, have a cell body measuring up to 135 microns. Granular neurons of the cerebellum, which are the smallest, measure 4 microns.
Large neurons have abundant rough endoplasmic reticulum (RER) which forms aggregates, the Nissl granules. These are easy to see with cellular stains such as the Nissl stain and H&E. If the axon is transsected, the RER disaggregates and the neuronal body balloons. The cytoplasm becomes smooth and the nucleus is displaced toward the periphery of the cell. This appearance, which is called central chromatolysis, is a reversible change that develops during repair of a neuron that has been disconnected from its target. Neurons contain numerous mitochondria that are needed for aerobic energy production. Deficiencies of mitochondrial enzymes affect primarily the brain and muscle. In the brain, mitochondrial disorders cause neuronal loss and present as encephalopathy, myoclonus, strokes, and other clinical disorders.
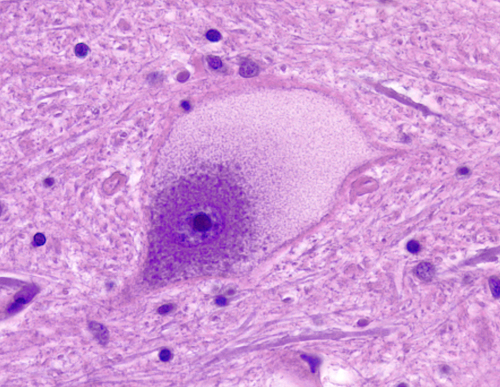
Hypoxia, ischemia, and hypoglycemia cause irreversible neuronal injury. Injured neurons shrink, become eosinophilic due to condensation of mitochondria, and their nuclei become pyknotic. Such neurons are referred to as anoxic neurons.
Neurons contain numerous lysosomes that are used for lifelong recycling of biomolecules and organelles. Deficiency of lysosomal enzymes causes unrecycled substrates of these enzymes to accumulate in lysosomes. Abnormal lysosomes gradually fill the cell body and processes, leading to destruction of neurons. There are many genetically transmitted lysosomal enzyme deficiencies (neuronal storage diseases).
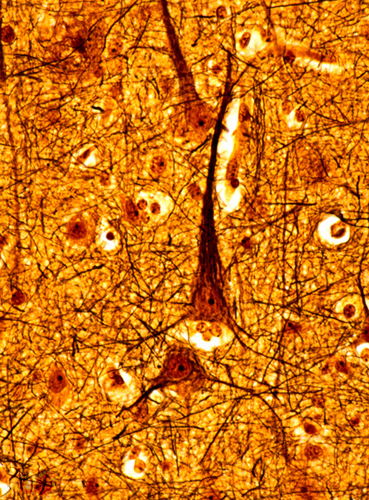
The neuronal perikaryon and processes contain longitudinally arranged 10 nm intermediate filaments (neurofilaments) and 20 to 26 nm tubules (neurotubules). Neurofilament proteins are chemically distinct from intermediate filament proteins of other cells. Based on their molecular weight, neurofilaments come in three sizes: light, medium, and heavy chains. When neurons are damaged, neurofilaments are released into the interstitial fluid and leak into the CSF and blood. Neurofilament protein levels are elevated in neurodegenerative disease and traumatic brain injury. Neurotubules are polymers of alpha and beta tubulin. Cross bridges made up of microtubule-associated protein (MAP) tau links neurotubules to one another and anchor them to other cellular structures. The neurotubules and neurofilaments form a lattice (cytoskeleton) which supports the structure of the axon and regulates axonal transport. Phosphorylation of tau influences the structural stability of axons and the speed of axoplasmic flow. Insoluble, hyperphosphorylated tau is deposited in the neuronal perikaryon in Alzheimer's disease and other neurodegenerative disorders (tauopathies). These deposits consist of paired helical filaments and form the neurofibrillary tangles (NFTs). CSF tau is elevated in Alzheimer’s disease.
The most important parts of the neuronal membrane are the synapses. Most synapses develop on thorn-like processes of dendrites, the dendritic spines. Each synapse consists of the presynaptic process (axon terminal), the synaptic cleft, and the postsynaptic process (part of a dendrite). The axon terminal contains neurotransmitters packaged in synaptic vesicles. The membrane of these vesicles contains special proteins, including synaptophysin and synapsins. Upon excitation, the synaptic vesicles fuse with the synaptic membrane and discharge their contents into the synaptic cleft. Contact of neurotransmitters with receptors on the postsynaptic membrane elicits cellular reactions that transmit the message to the postsynaptic neuron. The most important excitatory neurotransmitter is glutamate and the most important inhibitory neurotransmitter is gamma amino butyric acid (GABA).
The Hematoxylin and Eosin (H&E) stain is adequate for routine study of cellular details of neurons and glial cells, but does not stain the neuronal processes. Axons and dendrites are demonstrated best with silver stains in which ammoniacal silver is deposited on cytoskeletal components and then reduced to black metallic silver. The most commonly used silver stain is the Bielschowsky stain, which shows normal axons and dendrites and also reveals the lesions of Alzheimer's disease. Specific chemical components of nerve cells such as cytoskeletal proteins and synaptophysin can be demonstrated by immunohistochemical methods.
The generic term "degeneration" in reference to the cortex or other neuronal systems (e.g. striatonigral degeneration, cerebellar degeneration) means gradual neuronal atrophy leading to neuronal loss. Such a process characterizes most "neurodegenerative" diseases. In some of these diseases, such as Alzheimer's disease and Parkinson's disease, there are also specific histopathologic changes. In others, there is only neuronal loss, brain atrophy, and gliosis.
The term "neuronal plasticity" (meaning originally the ability to mold and change shape) refers to change or adaptation of neuronal function and structure dependent on activity. A clinical example of plasticity is the resumption of a function, e.g. speaking or control of swallowing, by another group of neurons when the neurons that originally performed this function are lost due to a stroke. The best known laboratory example of plasticity is long-term potentiation. The neuronal structure, especially the density of dendritic spines, changes as a result of neuronal activity. Neuronal plasticity is the basis of learning.