PARKINSON'S DISEASE
Parkinson's disease (PD) is the second most prevalent neurodegenerative disorder after Alzheimer's disease (AD) and the most common subcortical degenerative disease. It affects approximately 1-2% of individuals over the age of 60, with typical onset in one's sixties. PD is characterized by motor symptoms such as rigidity, resting tremors, slowness of movement, stooped posture, a shuffling gait, and balance issues, alongside nonmotor symptoms like a lack of facial expression, soft speech, loss of smell, mood disorders, dementia, sleep disturbances, and autonomic dysfunction, which includes constipation, cardiac arrhythmias, and low blood pressure. Notably, constipation affects between 24.6% and 63% of PD patients and may occur even before motor symptoms develop.
The vast majority of PD cases are sporadic, likely resulting from a combination of genetic and environmental influences. About 5-10% of patients have a hereditary form of PD, either autosomal dominant or recessive. The first identified autosomal dominant variant stems from point mutations or duplications and triplications of the α-synuclein gene (SNCA) located on chromosome 4q22. The protein α-synuclein is found in various organs and tissues. In the brain, it is primarily located in the neuronal cytosol and is attached also to the nuclear membrane and other cellular structures, including synapses. Although normal α-synuclein is soluble, mutations can lead it to form insoluble filaments that aggregate in the cytosol, in neuronal processes and glial cells. Alpha-synuclein plays a significant role in synaptic function, including the regulation of dopamine levels produced by the substantia nigra. PD may be caused by loss of function of normal α-synuclein or by toxic gain of function of its mutated forms. Additionally, large aggregates of it may crowd out cellular organelles, impair cellular functions and lead to neuronal death. The gene most frequently linked to autosomal recessive PD is Parkin, among several other genes that increase the risk of developing PD. Environmental toxins such as trichloroethylene, paraquat, and rotenone have also been implicated.
Patients with Gaucher disease (GD), particularly those with GBA1 mutations, exhibit a 20- to 30-fold increased risk of developing PD, and 7-10% of PD patients carry GBA1 mutations. The clinical and pathological presentations of PD in GD are similar to idiopathic PD but occur earlier with more pronounced cognitive deficits. The underlying mechanism linking GD to PD is unclear, but it is believed to involve lysosomal impairment, mitochondrial dysfunction, and endoplasmic reticulum stress, which ultimately impair the degradation of α-synuclein, leading to its accumulation.
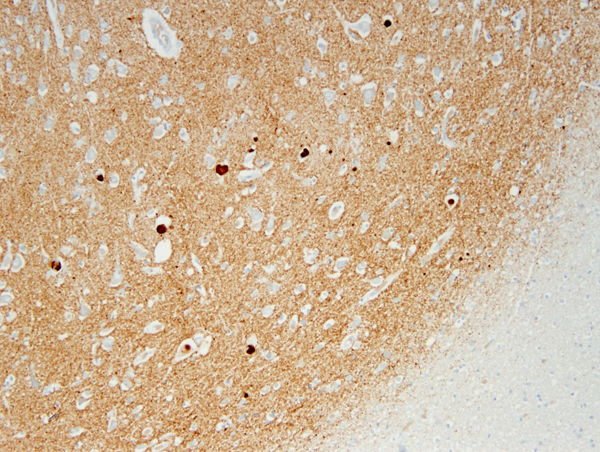
The key pathology in PD is α-synuclein accumulation. Large α-synuclein aggregates form round lamellated eosinophilic cytoplasmic inclusions, Lewy bodies (LBs), in the neuronal body, and fibrils, made of insoluble polymers of α-synuclein (Lewy neurites), are deposited in neuronal processes and in astrocytes and oligodendroglial cells.
This pathology primarily affects the dopamine-producing neurons of the substantia nigra (SN). These neurons are distinguished by their extensive and highly branched axons, numerous synapses, and high energy demands. The midportion of the SN, known as the zona compacta, is the first and most severely affected region. In advanced stages of PD, the loss of pigmented neurons results in a noticeable loss of pigment in the SN. As these dying neurons release neuromelanin, macrophages and astrocytes pick it up, with a small amount remaining free in the neuropil.
Dopamine, a crucial neurotransmitter, is synthesized by SN neurons from DOPA (which is also a precursor of melanin). Dopamine is then transported along the axons of these neurons to the striatum. The classic triad of symptoms in PD, rigidity, bradykinesia (slowness of movement), and resting tremor, corresponds to the degeneration of the dopaminergic nigrostriatal pathway and the resulting decrease in dopamine levels in the striatum.
Beyond the SN, abnormal deposits of α-synuclein protein and LBs are also found in various regions throughout the CNS, including the cerebral cortex, limbic system, and multiple extra-nigral neuronal groups such as the reticular activating system, locus coeruleus, dorsal motor nucleus of the vagus, nucleus basalis of Meynert, and olfactory nerves. Alpha-synuclein deposits are also found in the sympathetic and parasympathetic autonomic nervous system (ANS), including sympathetic ganglia, and myenteric plexus. This widespread distribution leads to impairment in the function of neurotransmitters like noradrenaline, serotonin, and acetylcholine.
This comprehensive pathology has only been fully understood in recent years, aided by the development of antibodies targeting α-synuclein, which make it easier to detect LBs and synuclein deposits outside the SN. Consequently, Parkinson's disease should be viewed as a systemic disorder affecting the entire CNS and ANS, not just the dopaminergic system. In fact, there is a proposal that some instances of PD might originate in the ANS, with α-synuclein spreading from the ANS to the CNS. This extensive involvement accounts for the diverse range of motor and non-motor symptoms seen in PD. Although LBs are characteristic of PD, they can also be found nonspecifically in various other neurodegenerative conditions.
AD and PD (and in a broader sense tauopathies and synucleinopathies) overlap clinically and pathologically. Extrapyramidal signs develop in AD patients, and dementia affects PD patients more frequently than age-matched controls. Also, LBs are found in the brains of AD patients, and a large proportion of PD patients have enough SPs and NFTs to qualify for the pathological diagnosis of AD. Until recently, dementia in PD had been attributed primarily to concomitant AD. Recent studies show that cognitive decline in PD correlates better with LBs present in the hippocampus, amygdala, and neocortex.
The treatment of PD consists mainly of L-dopa given with a DOPA decarboxylase inhibitor. An additional therapy for PD patients whose motor symptoms respond to but are not optimally controlled by medication and who do not have dementia is deep brain stimulation (DBS). In DBS, electrodes implanted into the subthalamic nucleus or inner part of the globus pallidus provide electrical stimulation to these and surrounding structures. This stimulation induces physiological and chemical changes that ameliorate some of the motor symptoms of PD by a poorly understood mechanism. Unilateral tremor and rigidity may also respond to stereotactic ablation of the contralateral globus pallidus, ventrolateral thalamus, or subthalamic nucleus.
DIFFUSE LEWY BODY DISEASE
Diffuse Lewy body disease (DLBD) (Lewy body dementia) is a sporadic neurodegenerative disease that is thought to be the second most common cause of dementia after AD. It combines the neurological manifestations of dementia and parkinsonism. Unlike AD, in which short-term memory is affected early, patients with DLBD present with fluctuating attention and cognition and visual hallucinations. Motor parkinsonian manifestations (bradykinesia, rigidity, and less frequently tremor) vary in severity and may appear later. DLBD patients also have depression, sleep disorders, and autonomic dysfunction. The brain in DLBD is not as atrophic as it is in AD and shows small, inconspicuous Lewy bodies in the neocortex, limbic system, and brainstem. A large proportion of DLBD patients also have AD pathology.
MPTP PARKINSONISM
The pyridine analogue MPTP (1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine) is taken up selectively by dopaminergic neurons. Its active compound, MPP (1-methyl-4-phenylpyridinium) inhibits mitochondrial function and induces cell death. Toxic damage of dopaminergic neurons causes parkinsonian symptoms. This was discovered when a drug addict accidentally injected himself with synthetic heroin made by himself, contaminated by MPTP. MPTP parkinsonism in humans and experimental animals resembles PD. Low dose intravenous infusion of the pesticide (and herbicide) rotenone, an inhibitor of complex I of the mitochondrial respiratory chain, causes degeneration of the SN and Lewy body-like inclusions in rats. The MPTP model, the rotenone experiments, and the ALS–Parkinson–dementia cases from Guam (see below) suggest that environmental neurotoxins play a role in the pathogenesis of PD.
GUAM ALS-PARKINSON-DEMENTIA COMPLEX (ALS/PDC)
A combination of amyotrophic lateral sclerosis, PD, and dementia was frequent in Guam and other Western Pacific locations. In ALS/PDC there is deposition of tau protein, α-synuclein, ubiquitin, TDP-43, and β-amyloid in the brain. NFTs are seen in the cortex and subcortical nuclei, including the SN; α-synuclein inclusions are present in the entorhinal cortex, amygdala, and SN; and TDP-43 inclusions are seen in neurons, oligodendrocytes, and astrocytes. There are few Alzheimer plaques. The appearance of this disease in successive generations implicated genetic factors. However, in recent years, the incidence of ALS/PDC has declined dramatically. Epidemiological work and animal experiments support the hypothesis that ALS/PDC is caused by cycasin and other neurotoxic compounds in the seeds of the cycad plant, which was used to make flour and was a staple in the local diet. The decline of ALS/PDC in recent years has been attributed to changes in diet and improved nutrition. ALS/PDC underlines the importance of environmental and nutritional factors in the pathogenesis of neurodegenerative diseases.
Parkinsonian syndromes may also develop in the course of other conditions that damage the SN, e.g., striatonigral degeneration, postencephalitic parkinsonism, manganese poisoning, carbon monoxide poisoning, hypoxic-ischemic encephalopathy, traumatic brain injury, and stroke.
Further Reading
- Houlden H, Singleton A B. The genetics and neuropathology of Parkinson's disease. Acta Neuropathol 2012;124:325–338.PubMed
- Okun MS. Deep-brain stimulation for Parkinson's disease. N Engl J Med 2012;367:1529–1538. PubMed
- Migdalska-Richards A, Schapira AH. The relationship between glucocerebrosidase mutations and Parkinson disease. J Neurochem 2016;139 Suppl 1:77-90. PubMed
- Lill CM. Genetics of Parkinson's disease. Mol Cell Probes. 2016;30:386-396.PubMed
- Diederich NJ, Uchihara T, Grillner S, and Goetz CG. The Evolution-Driven Signature of Parkinson’s Disease. Trends Neurosci 2020 Jul;43(7):475-492.PubMed
- Rehana K, Leake RK, Clark RN, Abbas M, et al. Current insights and assumptions on α‑synuclein in Lewy body disease. Acta Neuropathologica (2024) 148:18. PubMed
Updated: December, 2024